Get Your Reimbursement Guide
The individual who appears is for illustrative purposes. The person depicted is a model and not a healthcare professional.

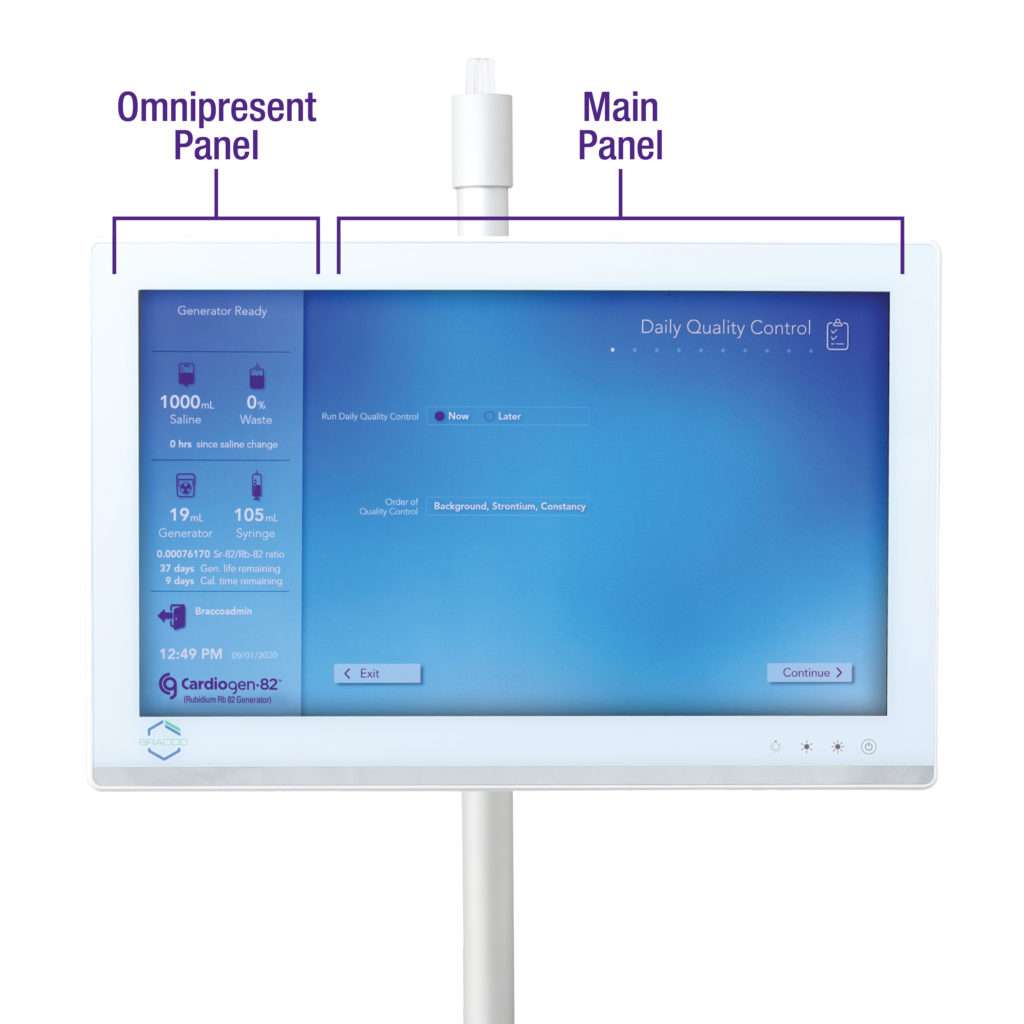
Designed with you in mind, the Model 1700 infusion system is focused on efficiency and ease of use – all while delivering the diagnostic confidence and safety you’ve come to depend on from the Cardiogen-82® generator.
Diagnostic confidence, safety, and efficiency plus the professional training, service, and expertise Bracco is known for.
Since 1989, Bracco has been building upon its legacy of leadership in cardiac positron emission tomography (PET) myocardial perfusion imaging (MPI) and the field of nuclear medicine. We were the first to invest heavily in cardiac PET MPI, and our continued commitment to the patients we serve each day is evident in the passion of our dedicated and experienced team. Join the growing number of world-class cardiac care facilities and teaching institutions that have discovered the benefits and clinical value of cardiac PET MPI with Cardiogen-82 generator.

ACCURACY

EFFICIENCY

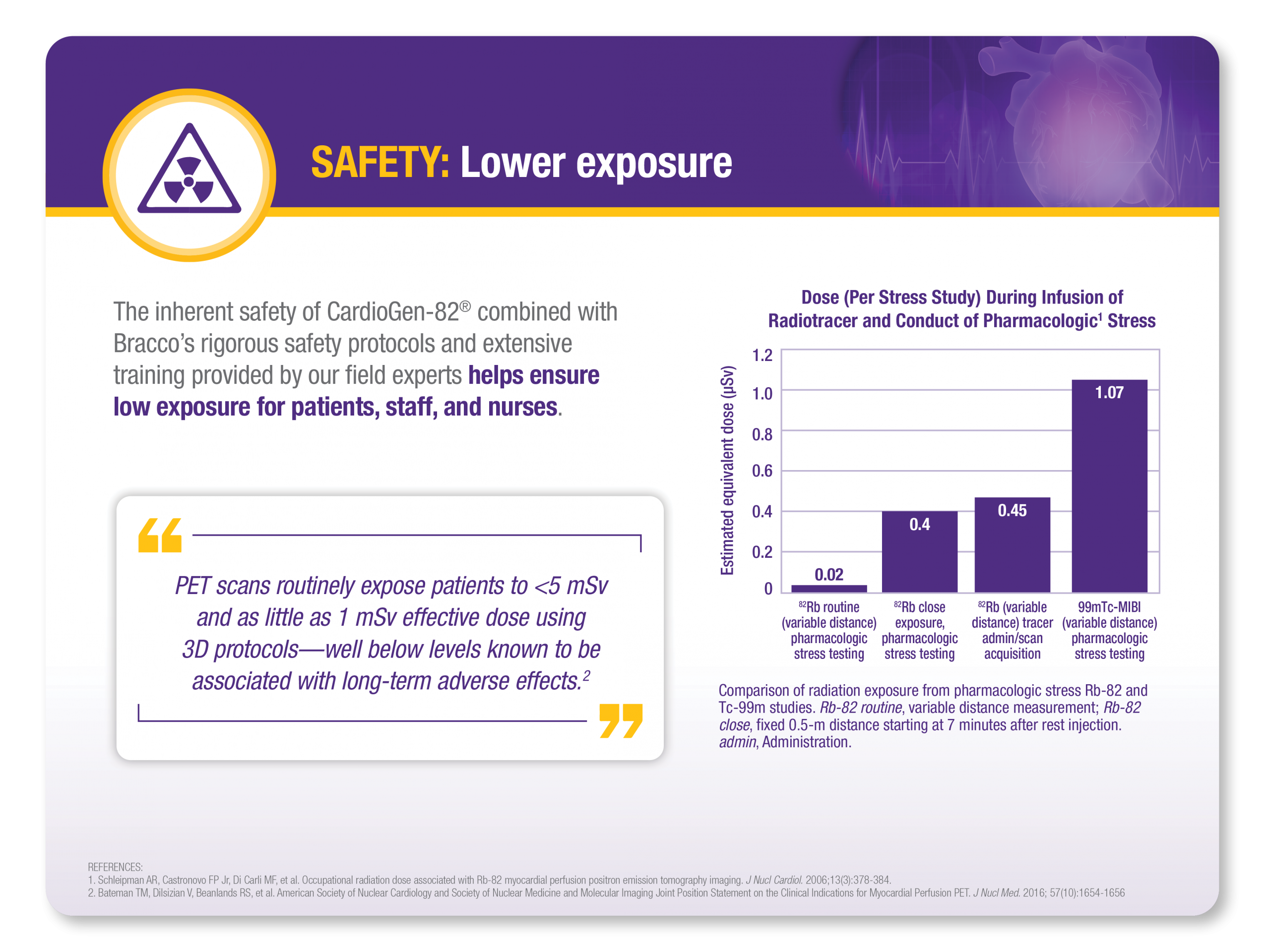
SAFETY
See what EXPERTS have to say.
See what the experts and patients of Cardiovascular Institute of the South in Houma, Louisiana have to say about the benefits of cardiac PET.
CARDIOGEN-82® (rubidium Rb 82 generator)
INDICATION CARDIOGEN-82 is a closed system used to produce rubidium Rb 82 chloride injection for intravenous administration and is indicated for Positron Emission Tomography (PET) imaging of the myocardium under rest or pharmacologic stress conditions to evaluate regional myocardial perfusion in adult patients with suspected or existing coronary artery disease. IMPORTANT SAFETY INFORMATION- Use only additive-free 0.9% Sodium Chloride Injection USP to elute the generator
- If an incorrect solution is used to elute CardioGen-82:
- Immediately stop the patient infusion;
- Evaluate the patient’s radiation absorbed dose and monitor for the effects of radiation to critical organs such as bone marrow;
- Permanently discontinue use of the affected generator.
- Record each generator eluate volume, including waste and test volumes, and keep a record of the cumulative eluate volume
- Strictly adhere to the generator eluate testing protocol, to minimize the risk of excess radiation exposure, including daily testing and additional testing at Alert Limits
- Stop using the generator if it reaches any of its Expiration Limits: 17 L for the generator’s cumulative eluate volume, or 42 days post generator calibration date, or an eluate Sr 82 level of 0.01 μCi /mCi Rb 82, or an eluate Sr 85 level of 0.1 μCi /mCi Rb 82